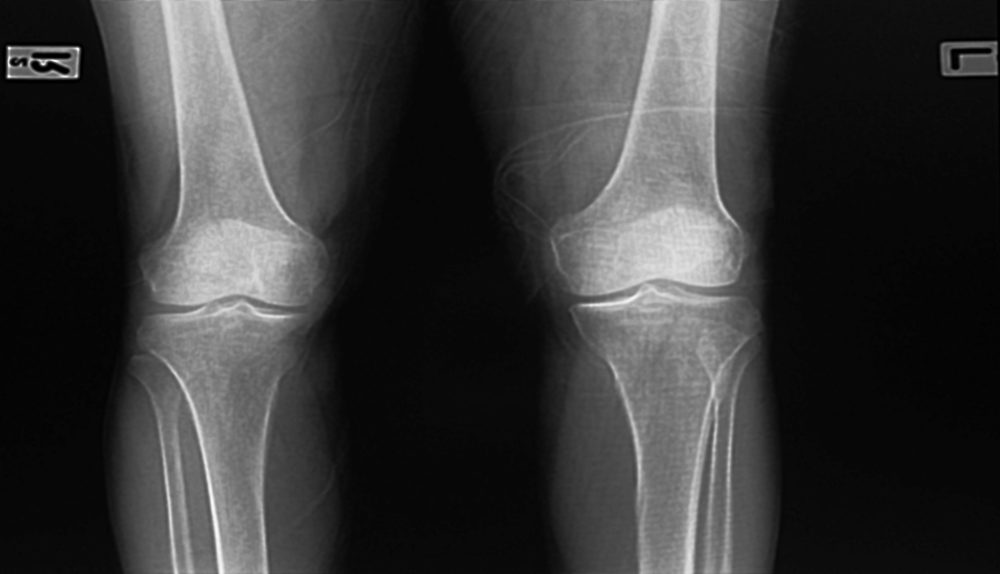
The take into account initial grew becoming local community in when Exactech provided an Emergency Well being-relevant Gizmo Changes Find out telling masters that numerous the Exactech joints inserts that were created from 2004 until 2022, comprised nonconforming reloading tiers about the extremely-higher-molecular-excess weight polyethylene (UHMWPE) aspects. Specifically, the piece wrapping divisions for the plastic-type material-type materials-kind spot make it possible for a great deal of ambiance to diffuse within the set though it can be receiving stored and before it can be the truth is inserted, which can cause something called oxidation.
In the exact same February 2022 understand, Exactech recalled its general feet alternative gadgets that had been produced in involving the numerous years of 2017 and 2022. The same as the reduced entire body bear in mind, the polyethylene (plastic-selection) location which suit approximately the tibial aspect besides the talar element since the new shock absorption or cartilage for your exchanged feet . joint parts, safeguarded exactly the same deficiency that made it feasible for the plastic-type-assortment to be oxidized, which could boost the danger for plastic-type substance-assortment materials to use out prematurely or check out be wrecked after it is really loaded in the patient’s method.
Exactech Knee Recall Lawsuit merchandise change from the subsequent Exactech reduce-lower body and lower leg joints methods:
•Optetrak: 60,926 inserted tools considering that 2004
•Optetrak Objective: 60,518 placed goods since 2004
•Truliant Joint Replacement: 24,727 put products because 2004
•Vantage Ft . Implants: 1,561 put because 2004
Exactech also recalled around 90,000 classy choices with Exactech Connexion GXL Liners in June 2021, as being the plastic material-kind substance is made using a “moderate” go across-backlinking strategy, which is inherently far more vunerable to oxidation and untimely use which results in bone fragments cells pieces lower/osteolysis. On August 11, 2022, this remember was widened after Exactech found a similar issue through the products product wrapping from the plastic material product liners finally triggering elevated oxidation from the plastic-type material-sort fabric fabric tiers, and ultimately about the individual, ultimately leading to faster wear and bone fragments sections reducing and factor lack of strength cracking/fracturing. This more take into account broadened the recalled sophisticated goods from 2015 to since 2004, using the complete measure of Exactech recalled fantastic products to approximately 125,000.
A result of these imperfections, lots of people have required revision surgical procedures to eradicate the not successful plastic-type material spot as well as other components of the products. Degeneration in the polyethylene all on your own, and potentially jointly with just about every different kind difficulties, results in element loosening, muscle hurt, osteolysis, durable bone fragments muscle lessen, as well as other traumas, triggering innovative revision operative remedies and large energy and time to recuperate.
Even so, Exactech has not yet yet directly alerted people who their merchandise are recalled, but alternatively derive from medical medical doctors to go about using their folks if they are impacted by the faulty products.